Jan. 5, 2018
UCalgary researcher leads Canada-wide clinical trial using anti-psychotic drug to treat ALS
If you took part in the ALS Ice Bucket Challenge, you may have wondered where the money raised by the millions of people who poured buckets of ice water over their heads went. Some of those funds are being invested in a University of Calgary research study investigating a potential drug treatment for amyotrophic lateral sclerosis (ALS) patients.
Dr. Lawrence Korngut, associate professor at the Cumming School of Medicine (CSM) and member of the Hotchkiss Brain Institute (HBI), is leading a clinical trial with nine hospital centres across Canada to recruit patients to confirm whether a well-known antipsychotic medication, pimozide, is safe and effective in stabilizing the progression of ALS over a six-month period.
The phase II clinical trial follows promising results from a collaboration between UCalgary and researchers at the University of Montreal. Their findings were recently published in JCI Insight.
Using the antipsychotic drug, pimozide, basic science researchers in Montreal were able to stabilize mobility in animal models — worms, fish and mice — born with the equivalent of the human form of ALS. Pimozide works especially well in preventing paralysis in fish with a genetic form of ALS by preserving neuromuscular function, a key finding with relevance to ALS in humans, which normally involves a progressive paralysis of the skeletal muscles.
“Pimozide has been well known for decades as a drug approved for treating certain types of psychiatric conditions, like schizophrenia, and it only costs nine cents per pill,” says Korngut. “Recent studies have shown genetic links between schizophrenia and ALS. We knew the next logical step was to test it on human volunteers — patients living with ALS.”
Korngut launched the first proof-of-principle clinical trial in 2015 with a small group of 25 ALS patients in Calgary. “We used a lower dose of pimozide than that used for other conditions, and now have preliminary proof that the lower dose may be useful in stabilizing ALS,” says Korngut.
The research team had an indication of the drug’s effectiveness after only six weeks. Loss of control of the thenar muscles, located in the palm of the hand between the thumb and index finger, is usually one of the first signs of ALS. For patients who took pimozide during the trial, this function remained stable. It is not yet known, however, whether pimozide has a curative effect, or whether it only preserves normal neuromuscular function to stabilize the disease, as a larger clinical trial is needed.
The discovery of a potential treatment marks an important turning point for ALS patients. There is currently no highly effective treatment for the disease, and it generally results in death within three to five years of diagnosis. However, Korngut emphasizes it’s too soon to draw firm conclusions about the safety and effectiveness of pimozide. “At this stage, people with ALS should not use this medication. We must confirm that it is useful and safe in the longer term. It is also important to be aware that pimozide is associated with significant side-effects. Therefore, it should only be prescribed in the context of a research study.”
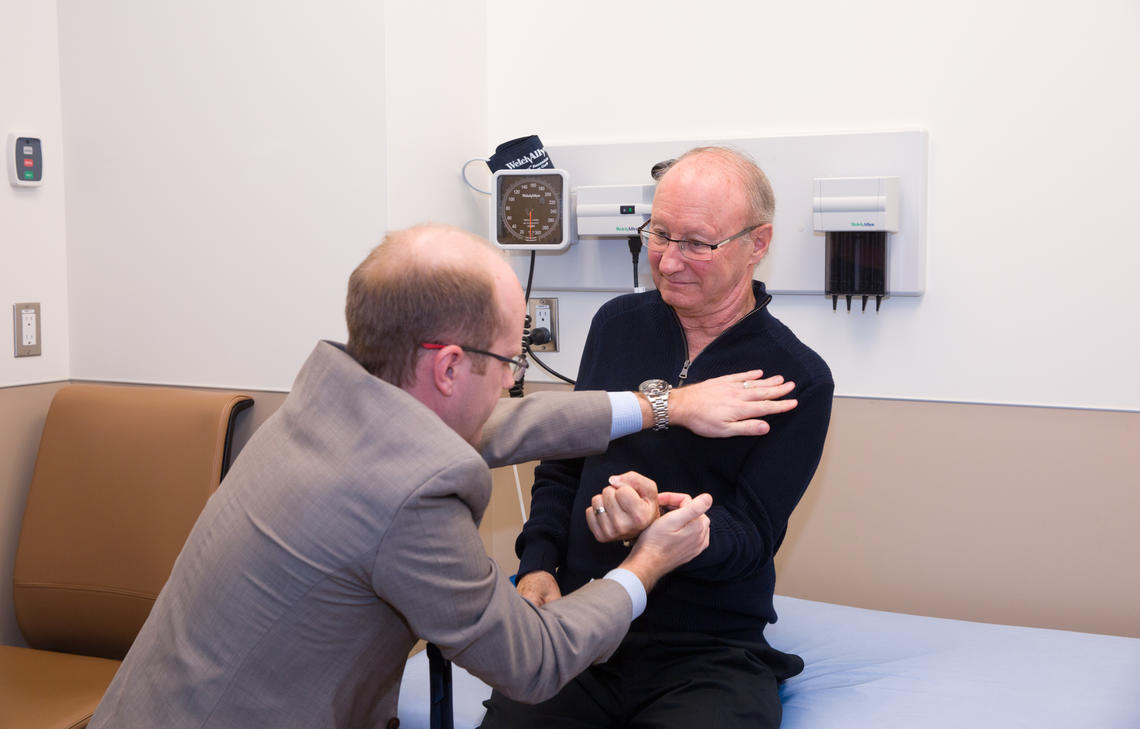
Dr. Lawrence Korngut examines his patient Cliff Barr as part of a clinical trial to treat ALS.
Riley Brandt, University of Calgary
ALS patients who are interested in participating in this study can contact pimozide2@ucalgary.ca, or find more information here.
Dr. Lawrence Korngut is an associate professor in the Department of Clinical Neurosciences at the CSM, a member of the HBI and director of the Calgary ALS/Motor Neuron Disease Clinic.
Funding for the first clinical trial for ALS was provided by the Quirk family of Calgary, the HBI, The Department of Clinical Neurosciences and the Clinical Research Unit at the University of Calgary.The clinical trial was conducted at South Health Campus in Calgary.
Funding for the phase II clinical trial comes from The Ice Bucket Challenge through a partnership between ALS Canada and Brain Canada.
Led by the HBI, Brain and Mental Health is one of six research strategies guiding the University of Calgary towards its Eyes High goals. The strategy provides a unifying direction for brain and mental health research at the university and positions researchers to unlock new discoveries and treatments for brain health in our community.
